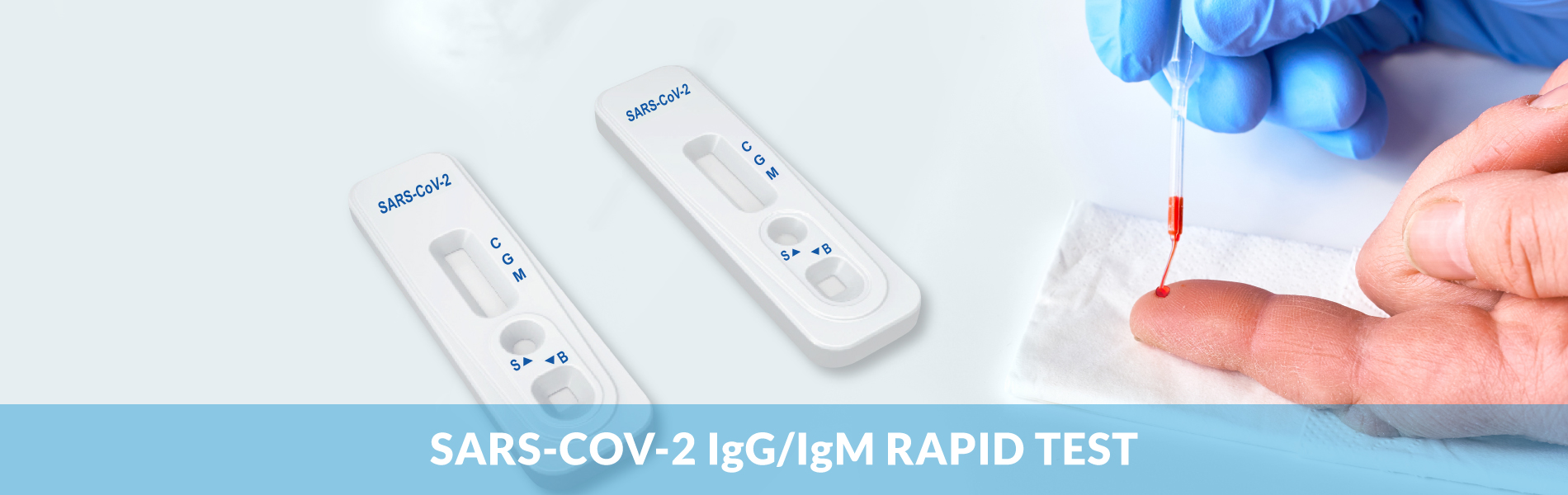
SARS-CoV-2 IgG/IgM Rapid Test
Detects IgG/IgM antibodies to SARS-CoV-2



Quick and accurate detection of IgG/IgM antibodies to SARS-CoV-2
A rapid test for the qualitative detection of IgM and IgG antibodies to the SARS-CoV-2(formerly known as 2019-nCoV) in serum, plasma, or whole and fingertip blood. For professional in vitro diagnostic use only.
Coronaviruses (CoV) are a large family of viruses that cause illness ranging from the common cold to more severe diseases such as Severe Acute Respiratory Syndrome. SARS-CoV-2 is a new strain that has not been previously identified in humans, and as the cause of an outbreak of respiratory illness first detected at the end of December, 2019.
In February 2020, the World Health Organization announced official names for both the virus and disease it causes as: SARS-CoV-2 and COVID-19, respectively.
How does the ACON SARS-Cov2 IgG/IgM rapid test work?
The SARS-COV-2 IgG/IgM Rapid Test is a qualitative membrane-based immunoassay for the detection of IgG and IgM antibodies to SARS-COV-2 in human serum, plasma, or whole blood. The membrane is pre-coated with anti-human IgM antibody and anti-human IgG antibody. During testing, SARS-COV-2 antibodies, if present in the specimen, will react with the SARS-COV-2 antigen-coated particles, which have been pre-coated on the test strip.
DOWNLOAD INFOAdditional Features
Accurate Results
98.5% Accuracy (96.9%-99.5%)* *95% Confidence Intervals
High Sensitivity and Specificity
Relative Sensitivity: 99.1% Relative Specificity: 98.2%
Multiple sample types
User Friendly Test Cassettes for Serum, Plasma, Whole Blood, Venous or Fingerstick samples
Additional Features
ACON Laboratories leads the way in providing high quality and innovative medical diagnostic products